Our goal was to develop a centralized, cloud-based platform that
empowers Regulatory Affairs (RA) teams in the MedTech industry
to digitize documentation, automate workflows, and reduce
time-to-market by streamlining compliance operations globally.
Strategic Vision: Digitizing Regulatory Complexity for MedTech
Excellence
-
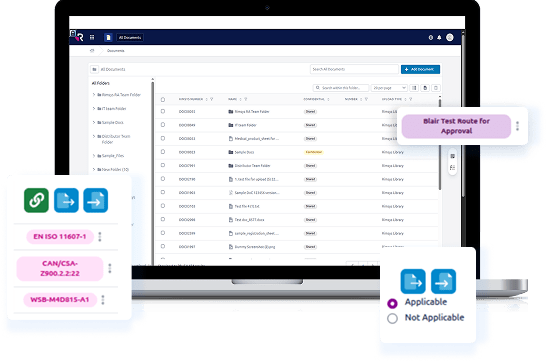
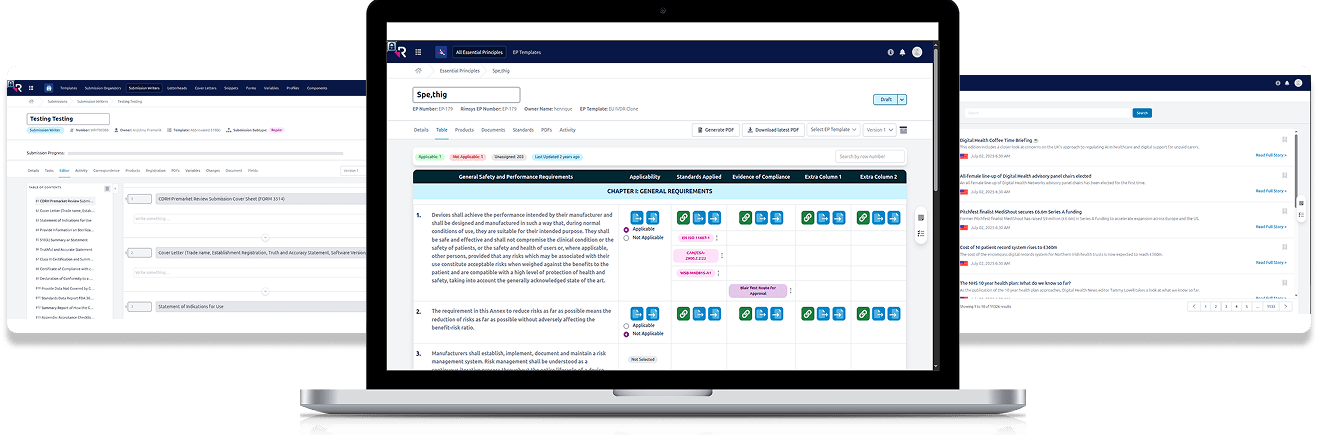
Centralized Information Architecture that serves as the
“single source of truth” for submission data.
-
Workflow Automation & Efficiency by logic-driven workflows.
-
Localization & Compliance Readiness with the integration of
country-specific regulatory entry requirements.
Guided by its vision to simplify complex enterprise workflows
through intelligent digital solutions, HeadBlocks partnered with
Rimsys to reimagine regulatory affairs for the MedTech industry.
The approach focused on building a unified, cloud-based platform
that digitizes documentation, centralizes compliance data, and
automates key regulatory operations.
By aligning with AWS best practices, implementing agile
development, and prioritizing user-centric design, HeadBlocks
delivered a scalable, secure solution that empowers RA teams to
reduce administrative overhead, ensure global compliance, and
bring products to market faster.